Osteoporosis Detection Ultrasonic Bone Densitometer KJ OSTEOKJ3700/3700S
Designed for pregnant women and infants, it tests bone density at multiple sites, has dual systems, and offers good performance and interaction.
Focusing on maternal and child bone density testing, it has multiple modes, dual systems, stable performance and easy operation.
Technical Specifications
Product Overview
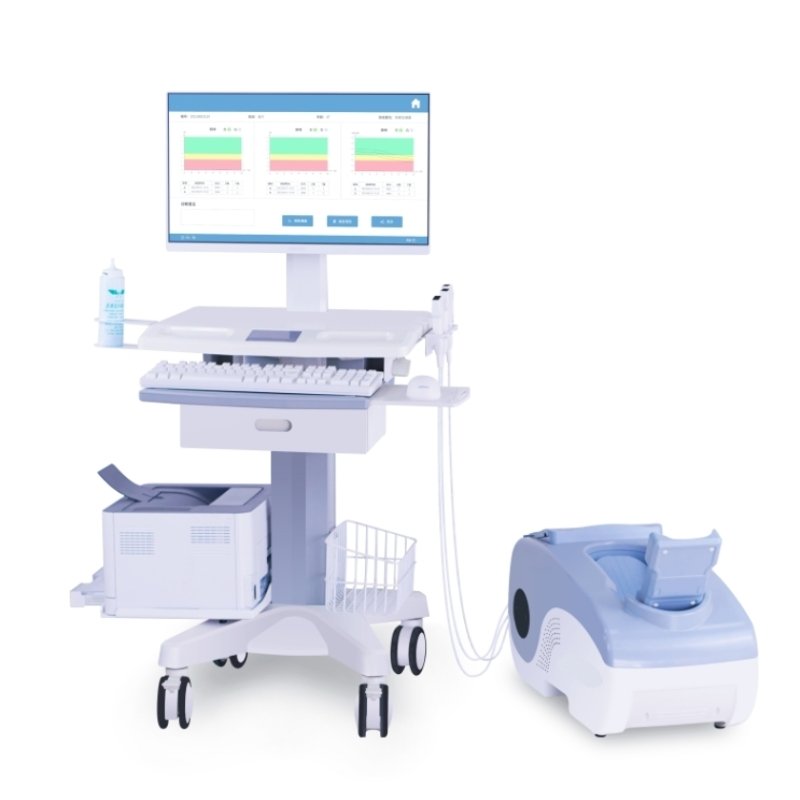
The OSTEO KJ3700/3700S Ultrasonic Bone Densitometer is a specialized device for bone density detection, dedicated to pregnant women and infants. It uses ultrasonic waves to measure the bone density of the calcaneus, tibia, and radius, providing comprehensive and accurate bone quality assessment. It supports dual - system operation of Android & Windows, enabling flexible and convenient use scenarios.
Detection Capabilities
Multi - site Measurement: It can measure six sites including the radius, tibia, and calcaneus, meeting the needs of different groups of people for bone density detection of various parts.
Specialized Reports: Provides dedicated reports for pregnant women and infants, facilitating targeted analysis and diagnosis by medical staff.
Detection Modes and Parameters
Calcaneus Mode: Can measure and display parameters such as SOS, BUA, Bone Quality Index (OI), T - Score, Z - Score, Adult Ratio (T - Ratio), Peer Ratio (Z - Ratio), Bone Biological Age, BMI, and Predicted Height.
Tibia/Radius Mode: Can measure and display parameters such as SOS, T - value, Z - value, Adult Ratio, Peer Ratio, Bone Biological Age, BQI, RRF, EOA, BMI, and Predicted Height.
Performance Indicators
Continuous Working Time: The host can work continuously for more than 8 hours, ensuring long - term and stable detection work.
Bluetooth Transmission: The Bluetooth transmission distance is not less than 5 meters, facilitating wireless data transmission between devices.
Liquid Ingress Protection: The tibia/radius ultrasonic probe meets the IPX7 liquid ingress protection level, ensuring the stability and durability of the probe in different environments.
Product Composition
- The product consists of the main unit, ultrasonic probe, auxiliary stand, calibration module, power cord, movable multi - hole socket, USB cable, positioning mark, software (Windows software version V1, Android software version V1), and optional components. The optional components include a color printer, computer (Windows platform, Android platform), table, tablet,and ultrasonic coupling agent.
Measurement Modes
- In the pre - measurement preparation interface, you can choose the "single - site mode" or "multi - site mode". The measurable sites include radius/left, radius/right, tibia/left, tibia/right, calcaneus/left, and calcaneus/right. After selecting the measurement site, click "Start Measurement".
Interface and Prompts
Tibia/Radius Mode: The "Measuring" interface will display the subject's information, real - time detection curve, ultrasonic probe parallelism, parallelism setting, and measurement results.
Calcaneus Mode: The "Measuring" interface will display the subject's information, real - time detection curve, measurement time, measurement progress bar, and prompt "Note: Do not move the position of the foot during measurement" to obtain more accurate data. When the subject is under 8 years old, the "Measuring" interface will play cartoons to soothe the subject.
Data and Interface
Windows Software: The multi - site ultrasonic bone densitometer Windows software supports obtaining the subject's information from the ID card reading device.
Android Software: The multi - site ultrasonic bone densitometer Android software supports obtaining reports by scanning the QR code with WeChat, printing reports through a network printer, connecting to the host through wireless Bluetooth, and obtaining data from the host.
Recommended External Configuration
- This product normally needs to be used with external devices such as computers and printers. Users can choose the optional accessories of this product. If users purchase and use them by themselves, the purchased devices should meet the requirements of GB9706.1 - 2020 standard and YY9706.102 - 2021 standard.








Reviews
There are no reviews yet.